Overview
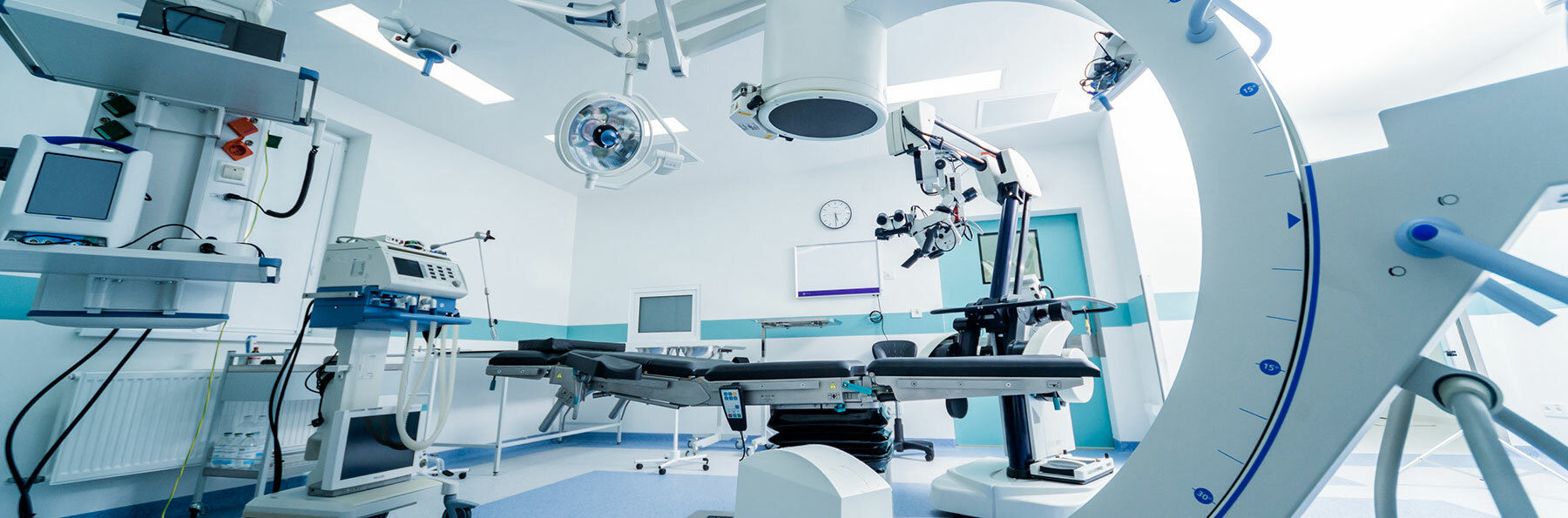
Medical device manufacturers need legal representation that understands clinical testing protocols and the processes for moving through Phase I and Phase II trials, all the way through FDA approval, in addition to labeling and advertising issues. Our attorneys monitor and track FDA developments, as well as developments in the medical device industry, including mergers and acquisitions and divestitures.
We work with large and venture-backed medical device manufacturers to meet complex regulatory requirements, to manage the risks associated with the marketing and distribution of devices, and to defend against claims in litigation.
We help clients focus on avoiding litigation and adverse regulatory actions. When litigation or adverse government regulatory action arises, however, we aggressively assert and defend our clients’ interests in every appropriate forum.
In medical device litigation, we understand the importance of drilling down to the central issues of the case at the beginning, in order to minimize the costly, prolonged discovery practices often associated with this type of litigation. Our familiarity with the laws and regulations applicable to medical devices, as well as data privacy and security, has helped achieve successful resolutions for our clients in these matters.
For example, by educating opposing counsel on the FDA 510(k) process, we limited discovery beyond the scope of the pertinent medical device issues alleged in a case. In another example, by having a thorough understanding of our client’s engineering and design drawings, the client’s witnesses were well-prepared at deposition.
Our experience with sensitive types of documents, such as the Design History File, allows us to efficiently review voluminous amounts of documents and not lose track of narrowing the scope of issues in a case. We have also developed a strong network of consultants and experts in various industries to assist with the evaluation and strategy of case throughout the course of litigation.
Our medical device clients range from small business owners to publicly traded companies, and we have represented clients in cases on a wide variety of devices, including:
- Gastrointestinal devices
- Pedicle screws used in spinal surgery
- Tracheostomy tubes
- Robotic surgeries
- Assistive and physical therapy devices
- Catheters
Our experience in these types of cases has also allowed us to become skilled in doctrine of FDA Preemption arguments, preparing witnesses noticed as 30(b)(6) deponents, and discovery control.
Professionals
- Partner
- Partner
- Paralegal
- Partner